Research Interests
I. Dynein Structure and RegulationCytoplasmic dynein is a molecular motor composed of a number of different proteins.1 It uses the energy derived from ATP hydrolysis to transport cellular cargo along microtubules, which are threadlike structures also constructed of proteins. In our research, we have focused on the synthesis and detailed study of individual subunits, particularly the light and intermediate chains, followed by studies to probe their interactions with each other and with other proteins. A detailed understanding of the structure and dynamics of dynein subunits, singly and as they interact with each other, creates the foundation for elucidating the molecular mechanisms of dynein assembly, regulation, and cargo binding. The figure below represents our current model for the configuration of the various subunits of the dynein motor. Since there is no structure available for the entire motor, the relative position of the subunits must be based on information gleaned from partial structures and spectral and thermodynamic evidence for interactions. New structural details of the dynein motor continue to emerge with intensive studies in our lab and a number of others. 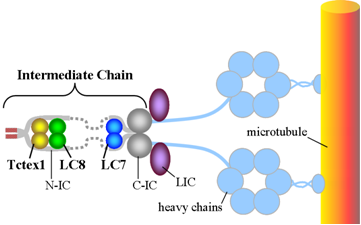 The dynein intermediate chain (IC) is central to the structure of the dynein motor.2 It is composed of two domains. The extended N-terminal domain (N-IC), is indicated by grey solid and dotted lines. The C-terminal domain (C-IC), which interacts with the heavy chain, forms a relatively ordered and compact beta propeller structure indicated by the grey globular shapes in the figure. Two copies of IC are present in every dynein motor, and the dimer is bridged by the three light chains LC8, LC7, and Tctex-1. In addition to the light chains, N-IC contains interaction motifs for several other proteins known to be integral to the function of dynein. These include p150Glued, the largest subunit of the 'dynein activator' dynactin, and other proteins such as LIS1 and the ZW10 subunit of the Rod RZZ complex. With its many interactions, N-IC appears to be the key modulator of dynein assembly and attachment to cargoes. With its extended structure, many interactions, and pivotal role in the function of a protein system, N-IC is representative of 'intrinsically disordered proteins'.3 The significance of this fascinating class of proteins has only recently been recognized. Because of their variability, the light chains have long been thought to regulate the activity of the motor, and current research supports a crucial role in this regard. Because they are known to interact with cellular components which are not unlikely cargo of the dynein motor, the light chains have also been thought to play a role in cargo attachment. However, recent structural and binding data from our lab and others suggests that the light chains cannot bind cargo proteins while a part of the dynein complex, and therefore cannot function as cargo adaptors. Our recent data strongly suggest that the tandem roles of dynein light chains LC8 and Tctex1 are to promote dynein assembly and the interaction with the cargo adaptor dynactin, and not to act as cargo adaptors themselves.4 Further, the light chains now appear to have diverse roles in the cellular economy distinct from their function in dynein, which may explain the perhaps misleading interactions with potential cargo molecules. II. Molecular Hub Proteins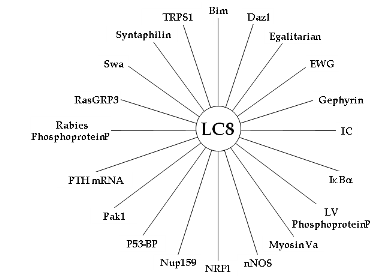
Dynein light chain LC8 in particular is known to interact with a very large number of proteins in addition to dynein components, as illustrated in the figure to the right. The LC8 sequence has been largely unchange throughout evolutionary history -- over 70% of the sequence of human LC8 is identical to LC8 from yeast -- indicating that these many interactions are critical enough to constrain the evolution of the protein. We have proposed that LC8 is a so-called 'hub' protein and functions as a dimerization facilitator in dynein and in all other complexes in which it participates.5 Projects in our lab will elucidate the binding thermodynamics and structural characterization of the two best-known proteins that interact with LC8: dynein intermediate chain (IC) and Swallow, a protein involved in localization of bicoid mRNA during Drosophila development. Swallow is the best-studied example of an LC8-interacting protein not associated with dynein. The mechanism and biological significance of LC8-promoted partner dimerization will be examined by both in vitro biophysical studies of representative protein segments and in vivo assessment of phenotypes of cells and organisms expressing designed variants of IC and swallow. These studies will provide a solid foundation for understanding the roles that LC8 plays in the variety of cellular systems in which it participates. These studies also have broader applicability, because LC8 can serve as a model for other hub proteins that interact with a large number of disordered partners. We envision both high-resolution structural studies of individual subunits with small peptide models of binding partners as well as thermodynamic characterization of the conformations of large domains of the disordered binding partners. III. Research MethodsIn our research we use a combination of NMR and X-ray crystallography and a battery of other biophysical methods, including isothermal titration calorimetry, circular dichroism, fluorescence, surface plasmon resonance, and mass spectrometry. In addition, a large part of our research is focused on developing methods for production of difficult to express and purify proteins, and for conducting in vivo assays. This research program therefore offers exciting opportunities for students and postdoctoral researchers who seek careers in structural biology/NMR spectroscopy at the interface of cell biology. IV. Further reading(1) Among the many recent overviews of dynein structure and biology see Vallee et al, 2004. (2) Our most recent work on the dynein intermediate chain is described here and here. (3) The properties of intrinsically disordered proteins were reviewed by Dunker et al., 2002. Those interested in Intrinsically Disordered Proteins should consider joining the IDP Subgroup of the Biophysical Society. (4) We discussed the interactions of dynein light chains in papers published in JMB (2006), JMB (2007), Biochemistry (2008), JMB (2008), and elsewhere. Please see the complete list of our papers here. (5) For information about protein 'hubs' see Han, et al and related citations. We proposed that LC8 is a hub here. |
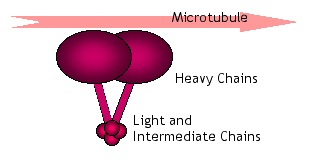 Transport by dynein motors is essential in several aspects of cell behavior, including cell migration and division, chromosome segregation, and transport of vesicles containing nutrients and other products. The protein subunits which come together to form the dynein motor are of several different types referred to as heavy, intermediate, and light chains, based on their relative size. The relatively invariant dynein heavy chain subunits bind to microtubules and contain the ATP sites that provide the motive force. The light and intermediate chain subunits are more variable in structure and play a more complex role in dynein activity.
Transport by dynein motors is essential in several aspects of cell behavior, including cell migration and division, chromosome segregation, and transport of vesicles containing nutrients and other products. The protein subunits which come together to form the dynein motor are of several different types referred to as heavy, intermediate, and light chains, based on their relative size. The relatively invariant dynein heavy chain subunits bind to microtubules and contain the ATP sites that provide the motive force. The light and intermediate chain subunits are more variable in structure and play a more complex role in dynein activity.